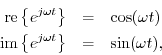That's the image I wish I had each time I state that vacuum polarization (due to the high charge density around the nucleus of an atom) creates a geometrical transform of the scalar quantum vacuum wave modes to a circular (spherical, given the DOF) orbital path of the bound electron, because a sinusoid is a circular function. This is what feeds energy to a bound electron to prevent it 'spiraling in' to the oppositely-charged proton(s) in the nucleus. At its ground state, the energy obtained from the quantum vacuum exactly equals the energy emitted via virtual photons (magnetism... which
all invariant-mass matter exhibits (usually diamagnetism, although certain electron valence configurations allow ferromagnetism to override the underlying diamagnetism)), as
Boyer,
NASA and
Haisch and Ibison showed,
You'll note the peak amplitude of the sinusoid is analogous to the radius of the circle, the peak-to-peak amplitude is analogous to the diameter of the circle, and the frequency of the sinusoid is analogous to the rotational rate of the circle. You'll further note the circumference of the circle is equal to 2 * pi radians, and the wavelength (wave period... the inverse of frequency) of a sinusoid is equal to 2 * pi radians, so the wavelength of the sinusoid is analogous to the circumference of the circle.
Why is this? Well, it's because a sinusoid
is a circle, spread out over space and time! It's a spiral. What we see on an oscilloscope is a sinusoid, but a truer representation would show a spiral over space and time... unfortunately, 4-D graphical displays don't exist.

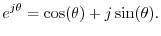
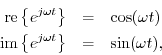
The above image shows the real (cosine... labeled 'Re' in the image above) and imaginary (sine... labeled 'Im' in the image above) components of a sinusoid.
So when viewed in line with its direction of travel, it will appear to be a circle, and when viewed orthogonal to its direction of travel, it appears to be a sinusoid, when in reality it's a spiral.
As an exercise, take a Slinky, stretch it out and shine a light behind it (it's best to do this with strong sunlight, as the light is nearly perfectly collimated, being from a distant 'point source'). You'll see its shadow is a sinusoid. So what we call sinusoids are really nothing more than shadows of a deeper reality. ;)
Why is the electron orbital spherical? The Higgs field causes invariant-mass matter (composed of nothing more than sinusoids of energy) to 'ping' back and forth off the Higgs field in a standing wave, which locks that energy to our frame of reference (which gives that energy the appearance of invariant mass and inertia). The electron (which has invariant mass) is orbiting in a spiral which is bound via the electromagnetic interaction to the nucleus (which has invariant mass), locked in time to our time frame by the Higgs field and thus forced to experience the passage of time, but free to move about the nucleus in 3-D space.
That's why Quantum Mechanics states that an electron orbit must consist of an integer number of de Broglie 'waves' (in actuality, an integer number of loops of a spiral), or it sets up a destructive-interference orbit... that's the 'why' of the quantization of quantum mechanics. That's also why it's so hard to pin down the electron's exact position in its orbit... it's spiraling in a circular orbit (imagine a spiral continually being pushed at a 90 degree angle due to the Lorentz force between electron and nucleus).
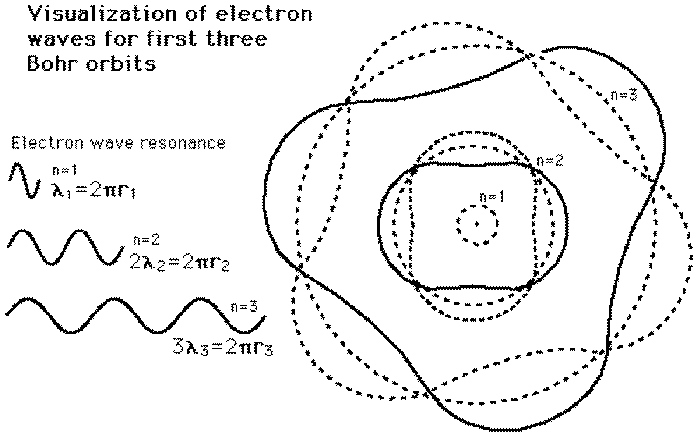
You'll note that electromagnetic energy, moving at
c (the speed of light),
experiences no time.
So the Higgs field causes energy to experience time, and the main difference between energy and invariant-mass matter is exactly that.